AP化学考点解析——元素周期律
来源:A加未来 时间:2020-07-27 17:12
元素周期律(Periodic law),指元素的性质随着元素的原子序数(即原子核外电子数或核电荷数)的递增呈周期性变化的规律。周期律的发现是化学系统化过程中的一个重要里程碑。今天A加未来小编就为大家分析在元素周期律中所涉及到的那些重要的AP化学考点,希望对大家有所帮助。
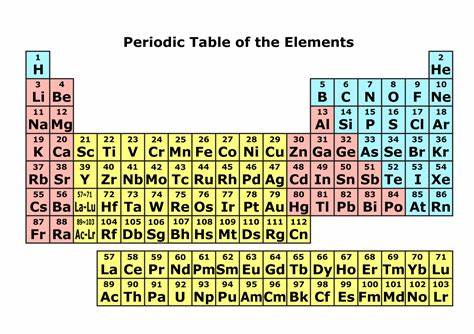
1)同周期电离能从左向右递增
Left to right
→more protons
→more positive charge in nucleus
→valence electrons more strongly attracted to nucleus
→more energy needed to remove electrons
2)同族电离能从上到下递减
Down a group
→more energy levels
→shielding effect
→less effective nuclear charge
→less attractions on valence electrons
→easier to remove
3)第二电离能大于第一电离能
When an electron is removed
→electron—electron repulsion decreases
→electrons are closer to nucleus
→increased attraction between electron and nucleus
→increased IE
4)当下一能量层移走电子时出现large jump(e.g.for Na,1st IE<<<2nd IE;for Mg,1st IE<2nd IE<<<3rd IE)
For Na,removing the second electron requires more energy because this electron is removed from its inner core(instead of its valence shell)
5)电负性在同一周期内从左向右递增,在同族内从上到下递减
It can be explained using the same explanation as atomic radius although the trend is opposite.
以上就是A加小编关于AP化学考点——元素周期律部分概念的解析,希望能够帮助大家更好的巩固这部分内容,并更好的应对AP考试。更多AP化学考试问题,欢迎随时咨询我们!
推荐课程
-
AP英语语言与写作课程辅导
你能正确分析一篇文章中的修辞手法吗?你能很好地论证...
-
AP课程周末同步课程
A加未来周末培训课程让学生通过每周末学习,定期强化...
-
AP物理1&2课程培训
1.AP physics 1&2课程内容 AP物理1,AP物理2课程的设置致力...
-
AP统计学辅导课程
课程介绍 AP统计学课程目的是培养学生学生关于收集、...
-
AP生物课程辅导培训
AP生物课程是针对没有接触过生物学科,或者接触甚少,...
推荐阅读
-
AP计算机科学考试内容解析
计算机科学作为第三次科技更名的开始,是现代社会发展......[详细]
-
AP物理1和2的区别对比,该考哪个?
自2015年AP考试改革后,AP物理B正式被取消,取而代之的是......[详细]
-
ap英语语言与写作难吗
我们一起来了解一下ap英语语言与写作的难度是多少吧。......[详细]
-
快速入门,AP微积分课程内容解析
微积分跟SAT和SAT2等考试不同,要求很强的基础知识储备......[详细]
-
AP计算机考点—解析Array和ArrayList的区别
在AP计算机考点学习中,很多同学们对于Array和ArrayList两......[详细]











 13699228024
13699228024  在线咨询
在线咨询 

